Unit 2: Organic Chemistry
Outcome:
MS3 Explore the nature and classification of organic compounds, and their uses in modern materials.
Resources:
Fractional Distillation Interactive
MS3 Explore the nature and classification of organic compounds, and their uses in modern materials.
Resources:
Fractional Distillation Interactive
Unit Test Outline
- Draw line diagrams for alkanes, alkenes and alkynes given the name
- Understand the intramolecular and intermolecular forces for hydrocarbons, including explaining what makes intermolecular forces stronger (i.e. longer chain, straighter chain, multiple bonds)
- Identify structural isomers of hydrocarbons
- Write and balance complete combustion reactions
- Identify products of incomplete combustion, and know why incomplete combustion may occur
- Explain why hydrocarbons make good fuels
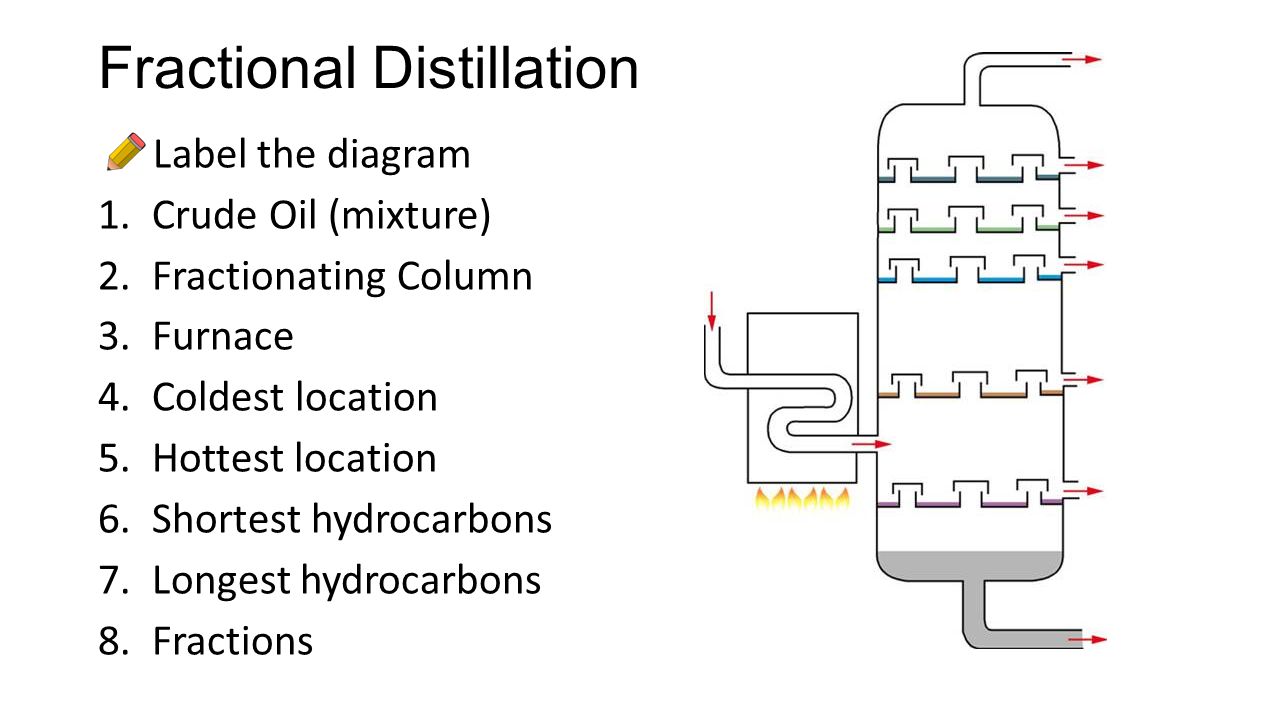
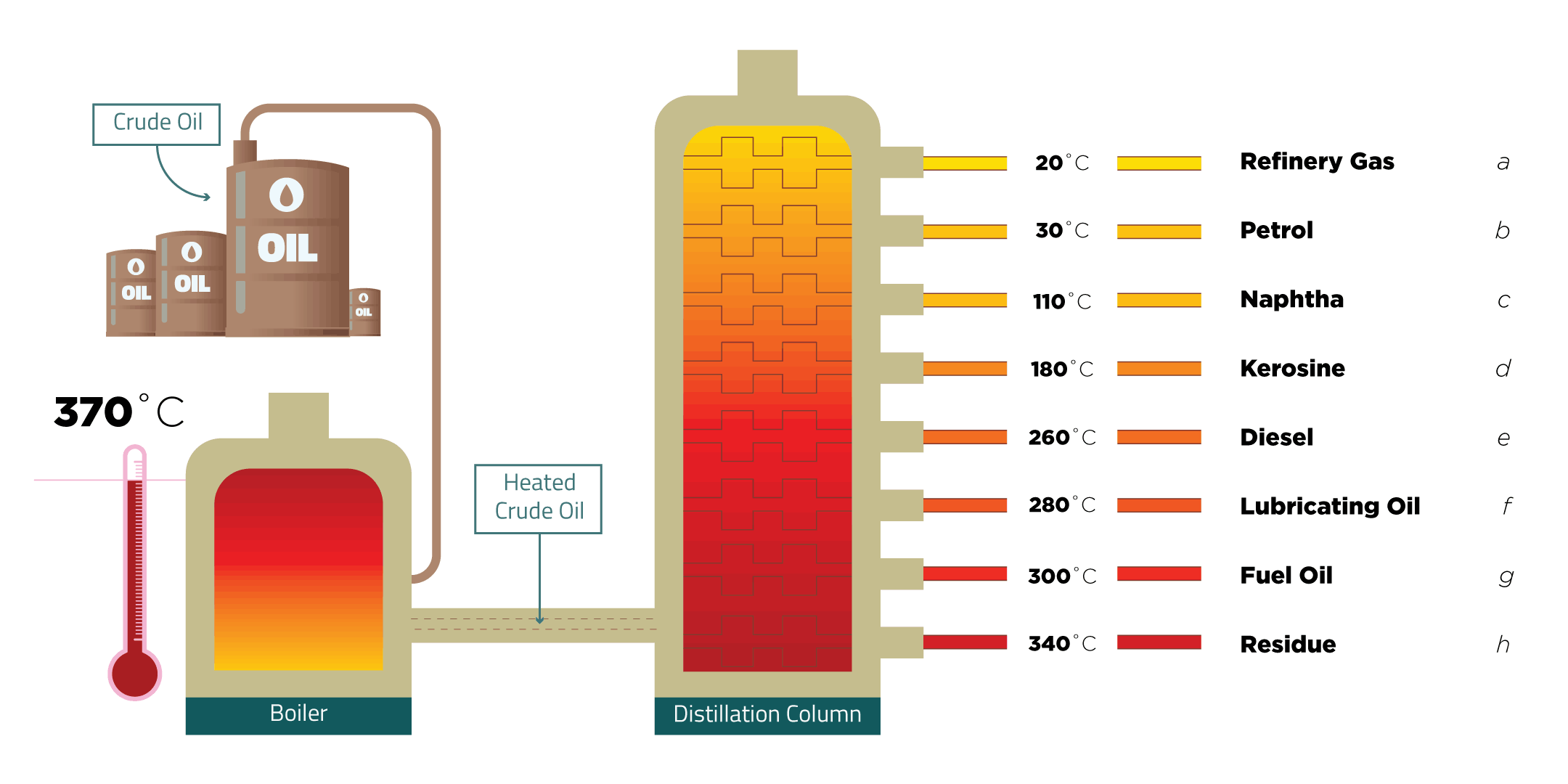
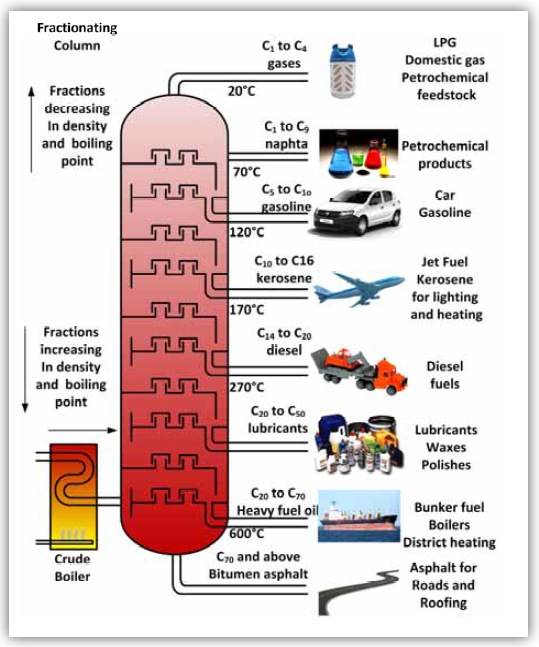
- Label a fractional distillation column and explain how the process of fractional distillation works
- Identify functional groups on a complex organic molecule
- Compare two compounds with functional groups to explain which will have stronger intermolecular forces
- Explain why compounds with short carbon chains and a functional group have different properties than a similar compounds with a longer carbon chain ((e.g. methanol vs pentanol)
- Draw line diagrams for alkanes, alkenes and alkynes given the name
- Understand the intramolecular and intermolecular forces for hydrocarbons, including explaining what makes intermolecular forces stronger (i.e. longer chain, straighter chain, multiple bonds)
- Identify structural isomers of hydrocarbons
- Write and balance complete combustion reactions
- Identify products of incomplete combustion, and know why incomplete combustion may occur
- Explain why hydrocarbons make good fuels
- Label a fractional distillation column and explain how the process of fractional distillation works
- Identify functional groups on a complex organic molecule
- Compare two compounds with functional groups to explain which will have stronger intermolecular forces
- Explain why compounds with short carbon chains and a functional group have different properties than a similar compounds with a longer carbon chain ((e.g. methanol vs pentanol)
| organic_chemistry_unit_notes.pdf | |
| File Size: | 507 kb |
| File Type: | |
|
| ||||||||||||
|
| ||||||||||||
| ica_structures_and_properties_of_isomers.pdf | |
| File Size: | 229 kb |
| File Type: | |
|
|
|